Digitalising how life‑science teams plan, execute & govern critical work.
The Digital Platform for Pharma & Biotech
One platform. Full visibility. Zero compliance gaps. Replace fragmented spreadsheets, siloed tools and paper-based workflows with a single GxP-ready digitalisation platform — purpose-built for regulated pharma and biotech organisations.
360° Platform visibility across every project | GxP Ready — built for regulated environments | 8–12 Weeks to full go-live
A sector under pressure to modernise — now
*AI-enabled performance metrics*
95%
of pharma projects experience schedule overruns due to poor planning tools – McKinsey & Co
40%
of compliance findings stem from inadequate documentation and audit trails
$2.6B
average cost to bring a new drug to market — every delay compounds losses
3X
faster project delivery reported by organisations using integrated digital platforms
Pharma project management is broken
“95% of pharma projects exceed budget or timeline” – McKinsey & Co, not because teams lack talent, but because they lack the right system.
Fragmented Systems
Teams operate across disconnected spreadsheets, emails and point tools — with no single source of truth for project data, decisions or approvals.
Compliance Risk
Manual tracking of GxP activities, audit trails and SOPs creates compliance exposure, slows regulatory submissions and leaves teams exposed at inspection.
Resource Blind Spots
Workforce visibility is limited. Planners cannot see cross-project conflicts, skill gaps or capacity constraints in real time — leading to delays and over-allocation.
Reporting Lag
Status reports take days to compile. By the time leadership sees the data, critical decisions are already delayed and cost overruns are already locked in.
One platform. Every capability you need.
goPLIMS supports end-to-end digitisation by replacing paper-based project control with a secure, integrated digital platform for planning, execution, reporting, and compliance.
Project Planning
Overall Planning
Create and manage project plans digitally with topic-wise actions, and dependencies in one place.
Work-flow Based task assignment
Use workflow-based task assignment to replace spreadsheets and paper trackers.
Multi-View Planning
Plan across multiple workstreams with calendar, list, and Kanban views for better prioritisation.
Meeting Actions & Follow-Up Tracking
Capture meeting actions, decisions, and follow-ups directly in the platform for traceable execution.
Resource Management
Role & Task Allocation
Assign tasks and responsibilities to individuals or cross-functional teams with clear ownership.
Workload Visibility
Track workload, status, and progress in real time to improve utilisation and reduce bottlenecks.
Team Collaboration
Coordinate global teams through centralised communication, attachments, and multilingual support.
Multi-Workstream Coordination
Monitor multiple streams or batches simultaneously to improve allocation and coordination.
Dashboards & Reporting
Customised Progress Reporting
Generate customised reports for project progress, actions, issues, and workstreams.
Real-Time Status Dashboards
Use real-time dashboards to provide visibility into milestones, risks, and readiness.
Reports Export & Sharing
Export reports to PDF or Excel for internal review and external sharing.
Centralised Management Reporting
Maintain centralised meeting reports and automatic updates for better management oversight.
Compliance & Audit
Audit Trail Management
Maintain a fully digital audit trail for actions, approvals, documents, and changes.
Controlled Document Management
Use controlled electronic document management with version control and secure access.
Digital Approvals & e-Signatures
Route records, forms, and approvals through digital workflows with e-signatures, create unique control copies.
Inspection Ready Traceability
Support inspection-ready traceability by keeping all project activity governed in one system.
Built for GxP. Ready for your next inspection.
Full Audit Trail
Every action, change and approval is logged with user identity, timestamp and reason — automatically. Complete, tamper-evident and always available at inspection.
GxP-Aligned Workflows
Configurable workflows enforce review and approval gates that satisfy GMP, GCP and GDP requirements — for every project type and activity across the organisation.
SOP & Document Control
Link controlled documents directly to tasks and projects. Always-current procedures at the point of work — with version control and change history built in.
Electronic Signatures (21 CFR Part 11)
Fully compliant e-signatures for all critical approvals and quality decisions — supporting 21 CFR Part 11 and EU Annex 11 requirements out of the box.
What inspection-readiness looks like with goPLIMS
Measurable outcomes from day one
Real improvements, backed by customer data from pharma and biotech teams using goPLIMS across clinical, manufacturing and QA programmes.
40%
Reduction in project reporting time — live dashboards replace manual status compilation
25%
Improvement in on-time project delivery across programmes
60%
Faster audit preparation — all evidence structured, searchable and exportable
35%
Reduction in resource planning effort through real-time capacity visibility
50%
Less time spent in status meetings — teams stay aligned through live dashboards
20%
Cost avoidance from proactive compliance management and reduced deviation incidents
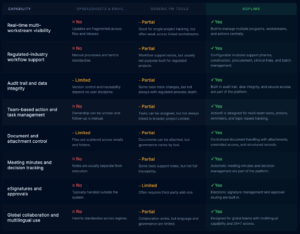
Why leading pharma teams choose goPLIMS
See how goPLIMS compares to the tools most pharma teams currently rely on.
We get you live without disrupting your operations
A proven, low-risk deployment path — tailored for regulated pharma environments where business continuity and compliance cannot be compromised.
01
2–3 Weeks
Discovery & Design
We map your workflows, system landscape and compliance requirements. Co-design the configuration with your team.
02
4–6 Weeks
Configure & Validate
Platform configured to your processes. CSV documentation produced. UAT and formal validation executed.
03
2–4 Weeks
Pilot & Go-Live
Pilot with a selected team or project type. Feedback loop incorporated. Full go-live with hypercare support from day one.
04
Ongoing
Adoption & Optimise
Dedicated Customer Success Manager. Quarterly business reviews. Continuous enhancement releases included in subscription.
Typical time to full go-live: 8–12 weeks. Pilot environments available within 2 weeks of contract signature.
The goPLIMS software suite for pharma digitalisation
Each module works standalone or as part of a fully integrated execution platform — configured for your workflows and compliance requirements.
ActionR
Action Register Management
Organise and manage all actions and tasks across workstreams. Customise workflows, automate tasks and visualise with Kanban boards.
RFI-M
Request for Information Management
Centrally raise all queries, review them with cross-functional teams, clarify all issues, review and resolve
ESignM
Electronic Signature Management
21 CFR Part 11 and EU Annex 11 compliant e-signatures. Structured approval sequences with full identity verification and audit trail.
DiscrepM
Discrepancy Management
Centrally manage exceptions, deviations and discrepancies. Root cause analysis, approval routing and full audit trail built in.
FormM
Electronic Form Management
Design custom e-forms, pre-approve, complete on any device, and integrate with document management for seamless paperless workflows.
EDocM
Electronic Document Management
Share, organise and collaborate on controlled documents. Manage versions centrally and define access at every level.
PunchM
Punchlist Management
Systematically identify, document and resolve snags, defects, incomplete work and issues on central register, track until closure with full traceability
ChangeM
Change Management
Structured change control with approval routing, impact assessment and full traceability — from initiation to closure.
TrackR
Central Online Tracker
Real-time visibility into status, milestones and dependencies across multiple workstreams. Customisable to any programme type.
DecisionM
Decision Management
Document collaborative decisions with rationale, approvals and audit trail — ensuring every key decision is traceable and auditable.
BatchM
Batch Management
Streamline and automate repetitive batch workflows. Centrally manage multiple batch processes with real-time status updates.
Low risk. High insight. Proven path to enterprise rollout.
Try goPLIMS with your real data and real projects — no long-term commitment required. Live in 2 weeks.

1 – This Week
Discovery Call
30-minute call to understand challenges and confirm pilot scope.
2 – Week 2
Pilot Agreement
Simple agreement signed. Sandbox provisioned. Templates selected.
3 – Weeks 2–3
Onboarding & Setup
Your team onboarded. Live data imported. Dashboards configured.
4 – Weeks 3–10
Pilot Execution
60-day pilot with full platform access and weekly CSM check-ins.
5 – Week 11+
Review & Roll-Out
Pilot debrief, ROI analysis and enterprise deployment plan.
One platform. Full visibility. Zero compliance gaps.
Let us digitalise the way your teams plan, execute and govern their most important work.