The goPLIMS Advantage
Built by someone who lived the problem for 30 years.
goPLIMS was not designed in a boardroom or by a software team guessing at pharma’s problems. It was built by a pharma project management expert who spent three decades watching critical projects struggle with the same preventable challenges — and decided to solve them once and for all.

The Origin Story
He didn’t set out to build software. He set out to fix a problem.
After three decades managing complex pharma projects — greenfield facilities, brownfield upgrades, technology transfers and commissioning programmes across three continents — Sundar Chellamani noticed the same pattern repeating, project after project, client after client.
Highly skilled teams were losing their most productive hours to chasing actions in email threads, manually compiling status reports from spreadsheets, attending meetings that existed only to compensate for the absence of a shared source of truth.
The tools available were either generic project management software that knew nothing of pharma compliance requirements — or industry-specific tools that were expensive, rigid and disconnected. Nothing bridged the gap between how pharma projects actually work and what technology could provide.
So Sundar built goPLIMS. Not as a software company building for pharma, but as a pharma expert building the tool his own teams needed. Every module reflects a real challenge he encountered. Every workflow was shaped by how C&Q, validation and operations teams actually operate on the ground.
“The majority of pharma projects are still run on spreadsheets, disconnected tools and emails. Teams spend their most productive hours in endless meetings, follow-ups and firefighting — not delivering. That is a problem with the system, not the people.”— Sundar Chellamani, Founder, Plimco Limited & goPLIMS
Sundar’s Journey
Pharma projects are still running on tools built for another era
Sundar spent 30 years watching these same three problems repeat across every project, every client, every site. They are not failures of people — they are failures of the systems those people are given.
Spreadsheets & Email as Project Systems
Critical project data — actions, changes, deviations, approvals — lives in email threads and spreadsheets with no version control, no audit trail and no single source of truth. Every status update requires manual compilation. Every handover risks information loss.
Disconnected Tools, Disconnected Teams
Engineering, CQV, quality, operations and document control each use different systems with no integration. Cross-functional visibility is impossible. Information is duplicated, conflicts are invisible until they become delays, and no one has the full picture at any moment.
Meetings, Follow-Ups & Firefighting
Without a live system managing actions and accountability, teams resort to meetings to share status, emails to chase follow-ups and phone calls to fight the fires that were invisible until they became urgent. Productive capacity is consumed by coordination overhead.
“Teams in pharma projects spend a disproportionate amount of their most productive time managing the management process — not delivering the project itself. That is the problem goPLIMS was built to eliminate.”
30% of project team time estimated lost to avoidable coordination overhead in manual execution environments
Plimco Limited & goPLIMS — better together
Two distinct organisations with one shared foundation: Sundar’s 30 years of pharma project experience. Together they offer something no competitor can replicate.
Plimco Limited
Plimco is Sundar’s specialist pharma project management consultancy — providing senior-level PM expertise to complex life science projects. Plimco does not just advise on process; it delivers projects, leading teams through the full lifecycle from design through to handover.
Greenfield & brownfield pharma facility projects
Commissioning, Qualification & Validation (C&Q/CQV)
Technology transfer & manufacturing scale-up
Digital transformation & Knowledge Management
GMP & regulatory compliance — ICH Q10, 21 CFR
Programme governance, PMO setup & team leadership
goPLIMS
goPLIMS is the purpose-built, GxP-ready integrated project execution & management platform that Sundar designed for pharma. It replaces spreadsheets, emails & disconnected tools with a single compliant system connecting all project workflows, teams & lifecycle stages.
Integrated action, document, change & decision management
21 CFR Part 11 compliant electronic signatures (ESignM)
Real-time dashboards & automated stakeholder reporting
Full audit trail — every action, decision & approval logged
Cloud SaaS — GxP-ready, lean & scalable
Configurable workflows without IT development overhead
No one else in the market offers this.
Generic software vendors understand technology but not pharma. Pharma consultancies understand the industry but have no platform. Plimco and goPLIMS together offer both — from one founder, one team, one integrated engagement model.
Implementation that speaks your language
When goPLIMS is implemented by Plimco, it is configured by people who understand GxP, C&Q, validation and pharma project methodology — not by generic software trainers working from a manual.
A platform shaped by the problems you face
Every goPLIMS module exists because Sundar encountered the exact workflow it solves — on a real project, with a real team. There are no features added for marketing value; everything reflects genuine operational need.
Continuity from advice to execution
With Plimco providing the PM leadership and goPLIMS providing the platform, strategy and execution are never disconnected. The same expertise that designs your approach also runs it — in the same system.
A partner invested in your outcome, not your licence
Sundar built goPLIMS to solve a problem he cared about. Plimco’s reputation is built on project delivery. Together, the incentive is always your success — not software subscription renewal.
Not just software. Not just consulting. Both.
When you engage Plimco and goPLIMS together, you receive an integrated offering that generic vendors and standalone consultancies simply cannot match.
A partner who already understands your world
Sundar has worked on the same type of projects your team delivers. He does not need to be educated on GMP, C&Q or what a punch list is. That context is built into every conversation, every implementation and every feature of goPLIMS.
A platform built from the inside out
goPLIMS was not designed by observing pharma from the outside. It was built from 30 years of working inside it. Every workflow, every module and every compliance control reflects how pharma teams actually operate — not how a software architect imagines they do.
Implementation with real PM methodology
A goPLIMS deployment supported by Plimco is implemented with full PM rigour — discovery workshops, workflow mapping, configuration governance, user acceptance and a structured go-live. Not a software handover and a user guide.
Compliance built in, not bolted on
21 CFR Part 11, GxP workflows, immutable audit trails and electronic signatures are core to goPLIMS architecture — not add-on modules. Plimco ensures the implementation reflects your specific regulatory context from day one.
Faster time-to-value than any generic solution
Because goPLIMS comes pre-configured with pharma templates, compliant workflows and industry-standard processes, and because the Plimco team does not need to learn your industry before they can help you, engagement to go-live is measured in weeks — not months.
A long-term partner invested in your performance
Plimco’s reputation is built on project delivery. goPLIMS grows as your organisation grows. The relationship is not transactional — it is a partnership between organisations that succeed only when your projects succeed.
30 years of experience distilled into one platform
Every goPLIMS feature represents a lesson from a real project, a real team and a real challenge — not a product roadmap decision made in isolation from the industry it serves.
30+
3
GxP
11
Specialisation Areas — Plimco
- Commissioning & Qualification (C&Q) for pharmaceutical facilities
- Validation — IQ, OQ, PQ and computer system validation (CSV)
- Technology transfer — development to commercial manufacturing
- Greenfield facility construction and brownfield upgrades
- Programme governance, PMO setup and project recovery
- Digital transformation and Knowledge Management (ICH Q10)
- GMP compliance, deviation management and CAPA
What That Experience Built — goPLIMS
- ActionR — meetings without structured action logs are wasted time
- DiscrepM — deviation investigations need full traceability from the start
- ChangeM — change without governed approval creates compliance risk
- EDocM — version-controlled documents are the baseline of GMP execution
- DecisionM — decisions made without records cannot be defended
- PunchM — defects managed in spreadsheets are incomplete
- ESignM — 21 CFR Part 11 compliance requires more than a PDF signature
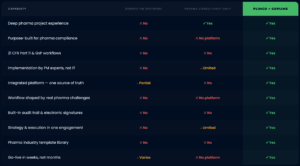
Why this combination is genuinely different
Compare what Plimco + goPLIMS provides against the alternatives your team is likely considering.
“I did not set out to build a software company. I set out to give pharma project teams back the time they were losing — the time that was going into spreadsheets, email chains and status meetings instead of into the work that actually delivers results for patients.”
Start with a conversation. Sundar will take it from there.
Every engagement begins with a discovery call — understanding your projects, your challenges and your goals before anything else is proposed.
Plimco + goPLIMS engagement:
Senior PM leadership from day one
Plimco provides experienced pharma PM expertise to lead your engagement — not junior account managers.
goPLIMS configured to your workflows
The platform is set up to reflect your processes, project types and compliance requirements — not a generic template.
Pharma templates & compliance controls pre-built
Industry-standard templates for C&Q, validation, change control and deviations are ready from go-live.
Dedicated Customer Success throughout
Ongoing support from a team that understands your project context and your platform configuration.
How the engagement works
30–45 minutes. We listen first. You share your projects, challenges and what you need. No pitch deck.
We propose the right combination of Plimco PM support and goPLIMS implementation for your specific situation.
Your processes mapped, goPLIMS configured to match, pharma templates and compliance controls set up.
Full 60-day pilot with your team on your live projects. Plimco provides PM guidance throughout.
Enterprise rollout, continuous improvement and an ongoing relationship — not a licence transaction.