BatchM – The Reliable Batch Management Software
The Reliable Batch Management Module
BatchM is a reliable module to support the automation and management of repetitive processes, lower cost, optimise resources and reduced waste, improve visibility interfaces with the operators and direct batch activity, enhance efficiency from initiation to monitoring and successful completion. The module is suitable for pharma, food, cosmetics etc.
Increase productivity and simplify complex tasks, centralize monitoring, resolve issues, and boost workflow efficiency.
Initiate – Monitor – Complete
Our Pillars
INITIATE
- Specify all dependencies
- Sequence all activities
- Detail the descriptions
- Specify Deadline
- Define the manpower
- Sequential activities
MONITOR
- Track live status online
- Monitor multiple streams centrally
- Resolve issues on time
- Monitor KPI’s
- Automated email reminders
REPEAT
- Stay in control
- Identify bottlenecks
- Effective scheduling
- Dynamic dashboard
- Generate system report
Challenges & Solutions
Dependency Coordination
Coordinating complex activities in sequence across diverse production elements.
Dependency Streamline
Streamline complex activities, sequence processes, and ensure efficient execution.
Real-Time Tracking
Challenge of ensuring real-time tracking and monitoring of multiple streams
Centralised Real-Time Monitoring
Enhancing visibility and process control
Bottleneck Resolution
Identifying and resolving bottlenecks promptly to maintain smooth production flow.
Bottlenecks Resolution
Visually identify bottlenecks, optimise workflow for consistency.
Inefficient Workforce Management
Managing workforce, adhering to deadlines, and handling unexpected disruptions effectively.
Dynamic Automation
Automates scheduling, adapts to disruptions, and streamline processes dynamically.
Revolutionize your team’s batch management with BatchM
– Seamlessly handle batch processes
– Set sequences for operations
– Alert operators
– Monitor progress on multiple batches simultaneously online
– Effectively manage bottlenecks and issues
– Optimize dependencies
– Enhance real-time oversight
– Empower diverse teams
– Collaborate on a central platform
Built for seamless collaborative Batch management
Streamline Process Initiation
Specify, sequence, describe, and manage for streamlined process initiation
Bottleneck Identification and Resolution
Identify and address issues for streamlined batch processes
Streamlined Process Sequencing
Sequence tasks for optimised and smooth processes
Dynamic Automation and Scheduling
Automate tasks, adjusting dynamically for optimized batch processes
Efficient Monitoring and Tracking
Monitor and track batches seamlessly for optimal efficiency
Enhanced Workforce Management
Manage workforce efficiently, ensuring productive and timely operations
Centralized Real-Time Monitoring
Centrally monitor operations in real-time for streamlined management
User-Friendly Workflow Enhancement
Enhance processes with a user-friendly approach for increased efficiency
Multi-lingual Selection
Options for multi language selection

Enhanced Efficiency

Centralised Oversight

Realtime Tracking

Streamlined Workflow
FAQ’S (Frequently Asked Questions)
BatchM by goPLIMS is a centralised, digital Batch Management platform that supports the automation and management of repetitive processes — from initiation through real-time monitoring to successful completion — across pharmaceutical, food, cosmetics, and other regulated production environments. Unlike spreadsheets or paper-based production logs — where batch activities are recorded after the fact, dependencies go untracked, operator visibility is limited, and bottlenecks are only discovered when they have already caused delays — BatchM provides a live, structured, and operator-facing platform that sequences every activity, assigns every responsibility, defines every dependency, and monitors every batch in real time from a centralised dashboard. Multiple batches across different business units and geographies can be managed simultaneously from a single platform, with each operator seeing their specific activities and each manager maintaining full programme-level visibility. The result is lower coordination costs, reduced waste, optimised resource utilisation, and a measurably more efficient production operation from the very first batch managed on the platform.
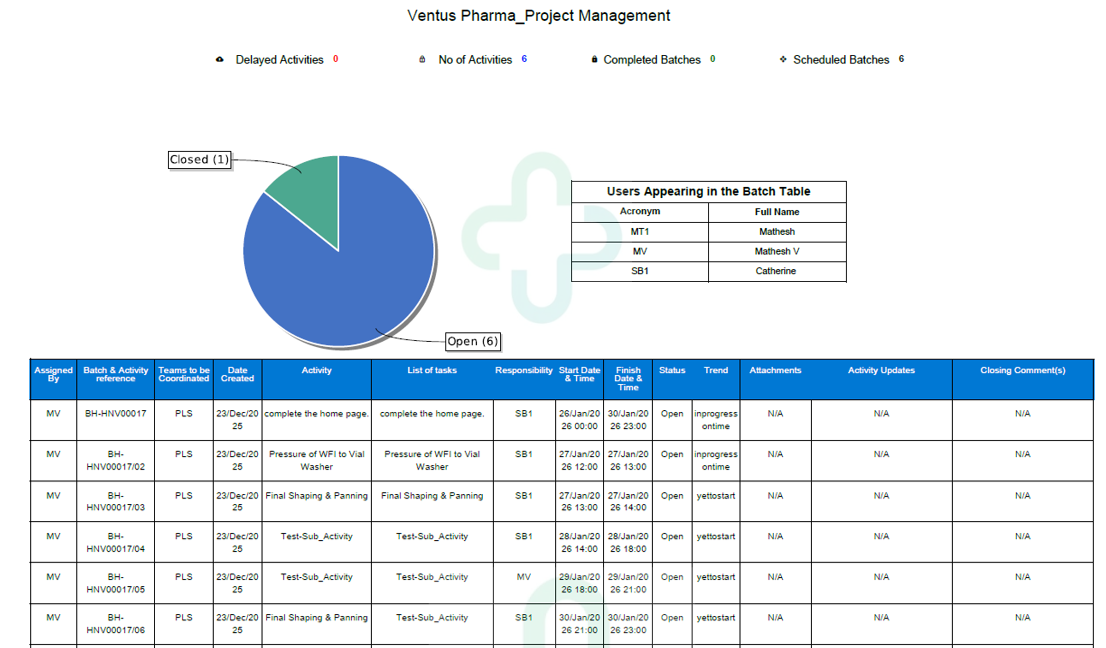
BatchM is built around three core pillars that reflect the complete operational lifecycle of any managed batch. Initiate: every batch begins with a structured setup — all dependencies between activities are specified, every task is sequenced in the correct operational order, detailed activity descriptions are provided to the operators, deadlines are defined for each step, manpower requirements are assigned, and sequential activity logic is configured to prevent steps from being executed out of order or before their prerequisites are met. This structured initiation removes the ambiguity and verbal instruction that create errors and rework in manual batch environments. Monitor: once the batch is live, all activity status is tracked in real time across the platform. Supervisors and managers can monitor multiple simultaneous batch streams from a single centralised dashboard, resolve issues and bottlenecks as they arise rather than after the fact, track KPIs against planned targets, and receive automated email reminders for approaching or overdue steps. Complete: the completion phase keeps the team in control as the batch moves toward closure — identifying remaining bottlenecks, enabling effective schedule management for the final activities, surfacing live status through dynamic dashboards, and generating formal batch completion reports that can be exported to PDF or Excel for records, review, or regulatory purposes.
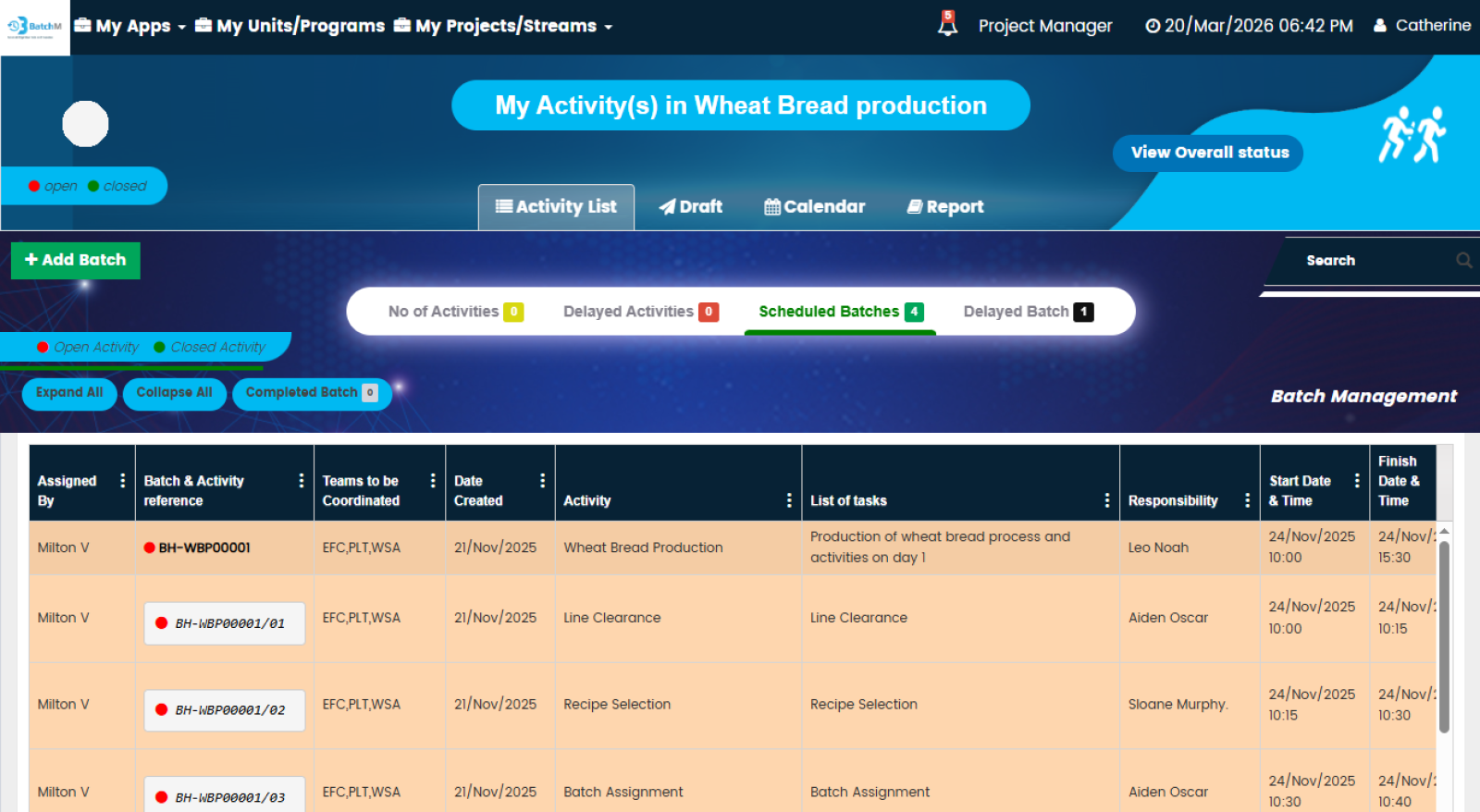
In regulated production environments — particularly pharmaceutical manufacturing, where executing a process step before its prerequisites are confirmed can invalidate an entire batch or trigger a GMP deviation — the management of activity dependencies is not an operational preference, it is a compliance requirement. BatchM addresses this directly through its dependency specification and sequential activity configuration at the Initiate stage. Every batch activity is mapped to its prerequisites, and the system is configured to prevent downstream steps from being released for execution until their upstream dependencies are completed and confirmed. This sequencing logic is enforced digitally — not dependent on verbal reminders, paper checklists, or operator memory — making it structurally impossible for activities to be executed out of sequence within a configured batch. For processes involving multiple parallel streams that must converge at defined synchronisation points — such as blending steps that require multiple separately prepared components to be ready simultaneously — BatchM’s dependency management ensures that convergence points are only reached when all contributing activities are complete. This eliminates the class of production errors caused by premature execution, missed prerequisites, and uncoordinated parallel workstreams that are common in manually managed batch environments.
BatchM is designed with the specific governance and traceability requirements of pharmaceutical batch management in mind. In GMP-regulated pharmaceutical production, every batch must be executed in accordance with approved procedures, with a complete and accurate record of who performed each activity, when it was performed.. BatchM’s structured activity sequencing, operator assignment, and real-time status tracking provide the operational framework to execute batches consistently and in compliance with approved procedures. Each activity is assigned to a named operator or team, ensuring that responsibility is explicit and traceable. Deadlines are defined at the activity level, with automated reminders ensuring that time-critical steps are not missed. The system generates batch completion reports that can be exported and retained as part of the production record. For pharmaceutical organisations transitioning from paper-based batch records to a digital batch management environment, BatchM provides a structured, governed, and scalable platform that supports traceability from day one while delivering the efficiency gains of a digitalised process.
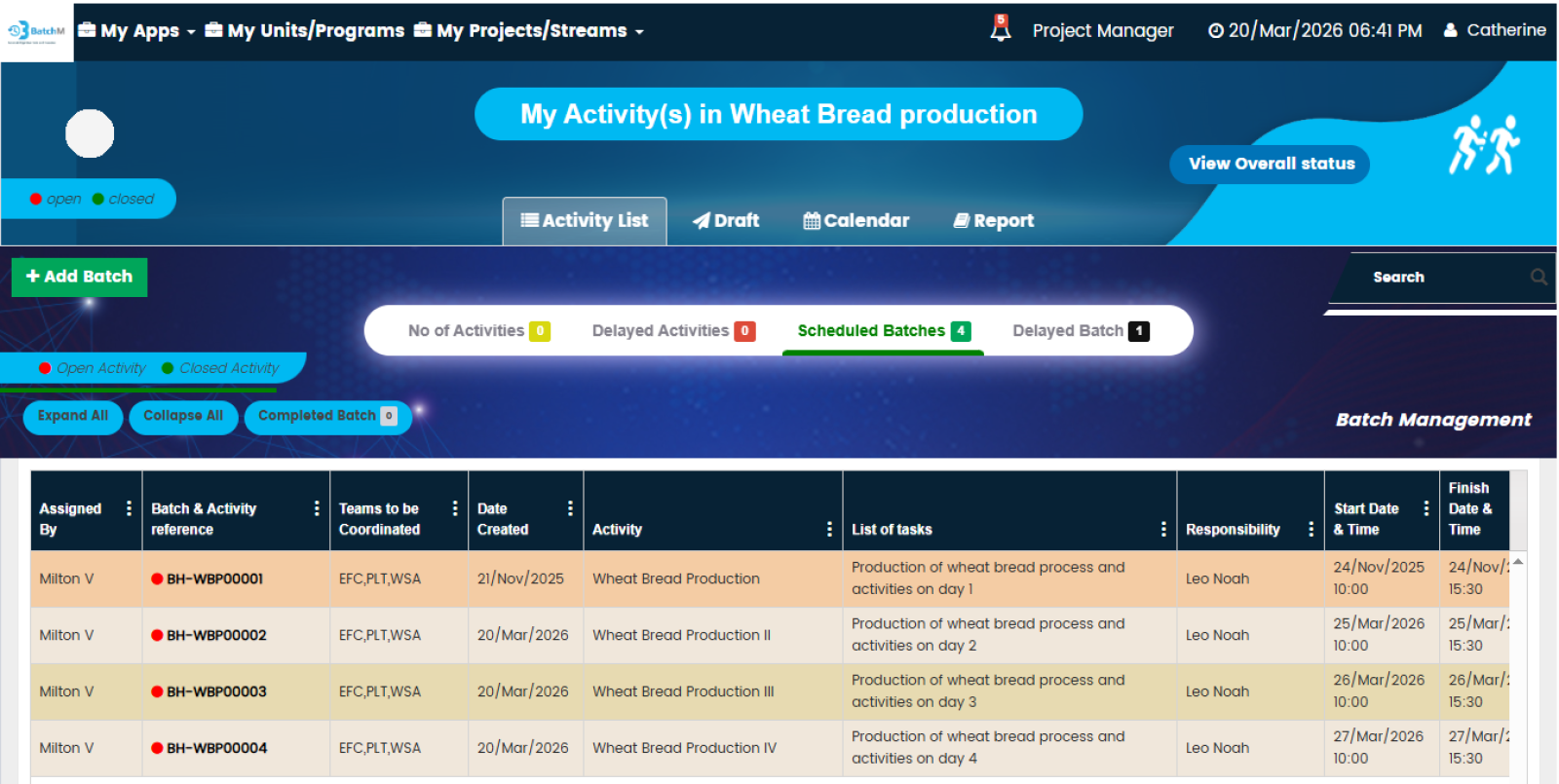
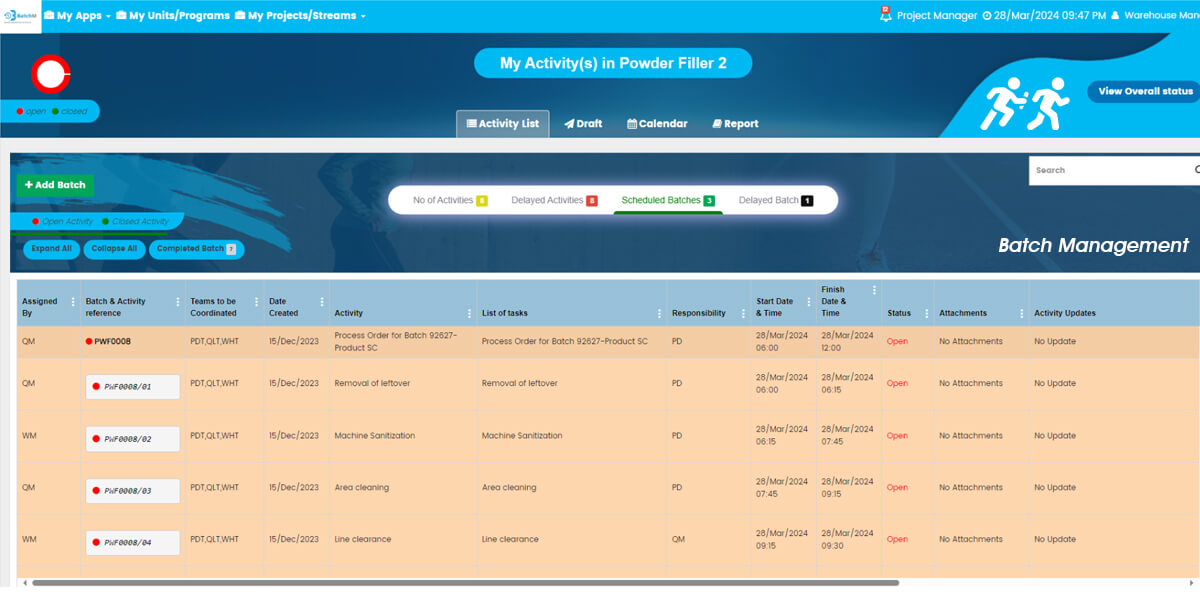
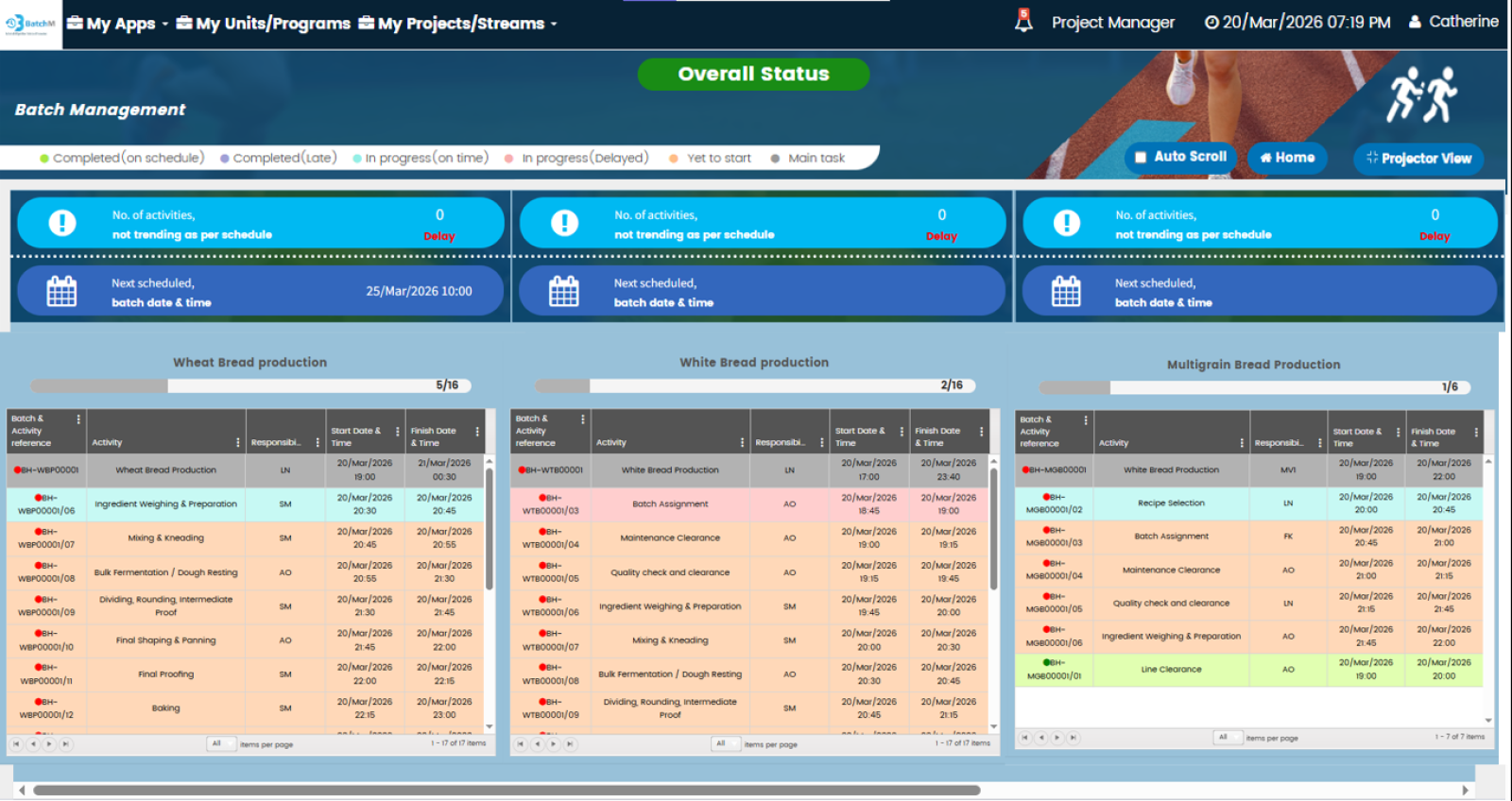
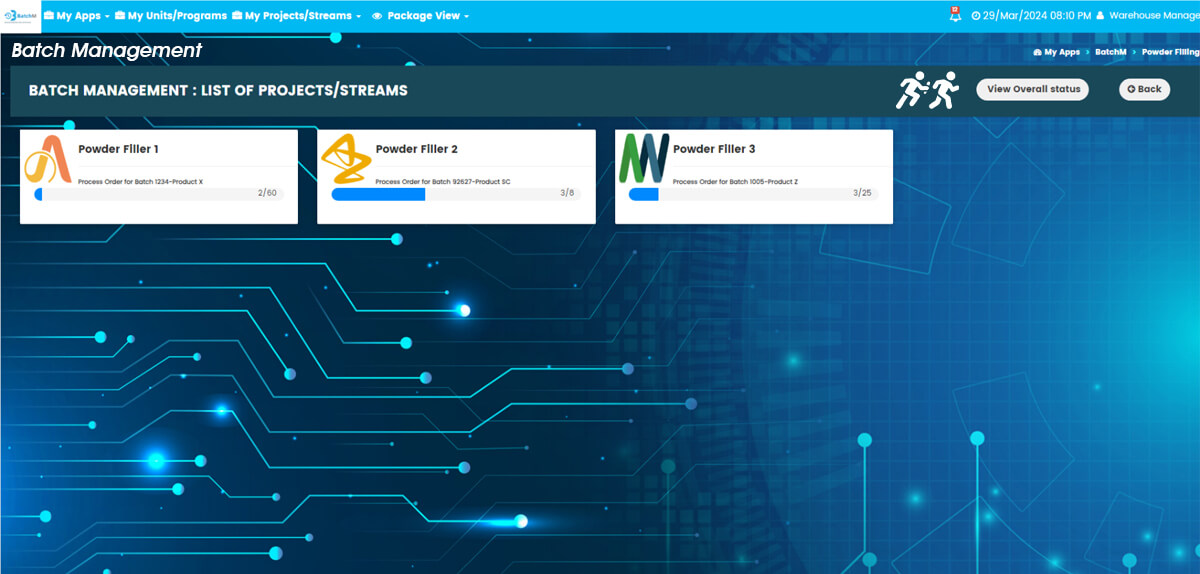
One of BatchM’s most powerful operational capabilities is its ability to manage and monitor multiple concurrent batch streams — across different products, production lines, business units, or even different geographic sites — from a single, centralised, real-time dashboard. In large pharmaceutical, food, or cosmetics manufacturing organisations running multiple batch programmes simultaneously, the traditional approach of managing each batch independently — with separate spreadsheets, paper logs, or disconnected systems — creates a fragmented visibility problem: supervisors and production managers cannot see the overall status of all active batches at once, making it impossible to identify resource conflicts, prioritise interventions, or report programme status accurately to management. BatchM’s Overall Batch Summary view resolves this by presenting the live status of every active batch — across all streams, all business units, and all locations — in a single consolidated dashboard, updated in real time. Programme Directors and Operations Managers can see at a glance which batches are on track, which are at risk, and which require immediate intervention — without navigating multiple separate systems or waiting for manual status updates from site teams.
Bottlenecks are one of the most costly and persistent problems in batch production management — and they are most damaging when they are identified late, after delays have already cascaded through the downstream schedule. BatchM is specifically designed to surface bottlenecks as early as possible and to equip supervisors and managers with the tools to resolve them before they affect batch completion. During the Monitor phase, BatchM provides real-time visibility of every activity’s status against its planned deadline — making it immediately apparent when a step is running late, when an operator is overloaded, or when a dependency is at risk of delaying a downstream activity. Automated email reminders are triggered as deadlines approach and when steps become overdue, ensuring that responsible parties are proactively alerted rather than reactively informed. The dynamic dashboard provides a visual representation of batch progress that makes bottlenecks and at-risk activities immediately identifiable without requiring manual analysis of status reports. The Collapse All and Expand All features allow supervisors to toggle between a high-level programme overview and a granular activity-level view in a single click — enabling rapid triage of where attention is most needed across a complex multi-batch environment.
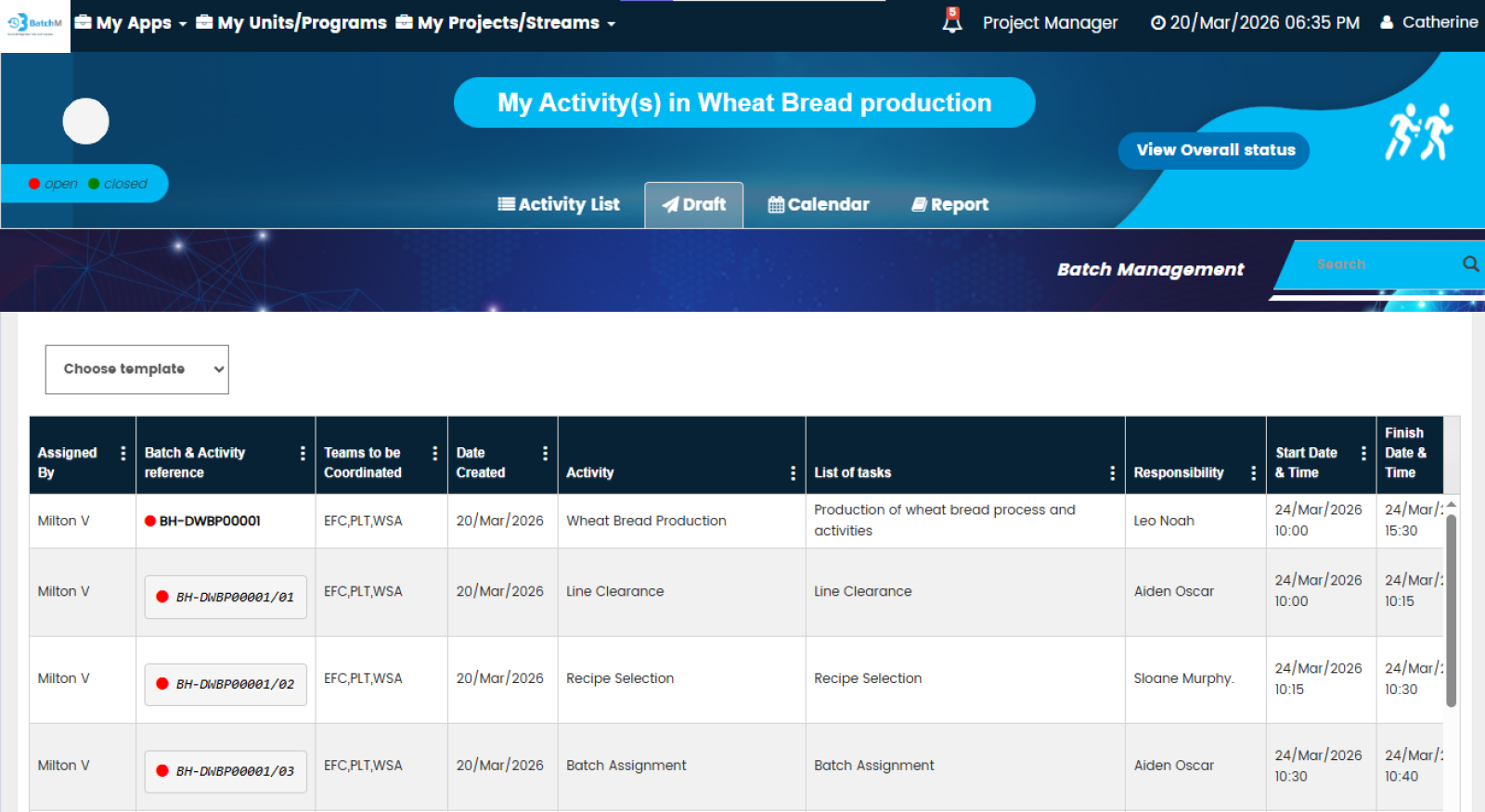
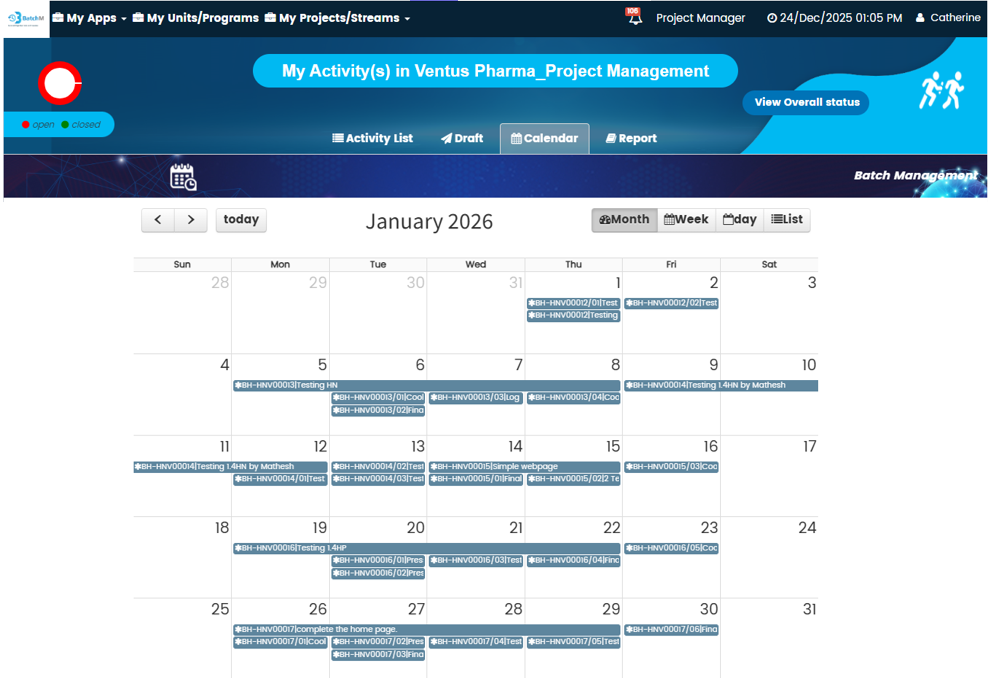
BatchM provides a set of purpose-built views that give different users exactly the level of detail and perspective they need to manage their role in the batch process effectively. The Overall Batch Summary provides a programme-level dashboard showing the live status of all active batches simultaneously — ideal for Production Managers, Operations Directors, and senior leadership who need an instant, consolidated picture of production health without navigating individual batch records. The Activity List View displays all sub-activities within a batch in a structured, sortable list — giving supervisors and operators a clear, prioritised view of every task and its status within a specific batch. The Calendar View maps all batch activities and completion deadlines onto a calendar, enabling planning teams and schedulers to identify deadline clusters, resource conflicts, and gaps in the production schedule in advance. The Collapse All and Expand All features enable users to rapidly switch between a summary batch view and a full granular activity detail view — making it easy to get both the overview and the detail without navigating multiple screens. The Draft Tab allows batch configurations to be saved and adjusted — with dates, responsibilities, and sequences modifiable — before the batch is formally initiated, ensuring that every batch is correctly set up before it goes live.
Effective batch management depends not just on defining what needs to be done, but on ensuring that the right people know exactly what they are responsible for and when. BatchM supports this through its operator-facing interface and structured workforce management capabilities. At the Initiate stage, manpower requirements are defined for each batch and each activity is assigned to a named operator or team — ensuring that every step has a clearly identified responsible party before the batch begins. Operators access BatchM directly and see exactly which activities are assigned to them, in what sequence, with what deadline — removing the ambiguity and verbal instruction chains that create errors and missed steps in manually managed environments. Automated email reminders notify operators when activities are approaching their deadline or have become overdue, ensuring that the pace of the batch is maintained without supervisors having to manually chase every step. For operations spanning multiple shifts, departments, or contractor teams, BatchM ensures continuity of information — incoming shift teams can see the exact status of every batch activity without relying on handover notes or verbal briefings. Multi-language capability allows operators working in different languages to interact with the platform in their preferred language — particularly valuable in multinational manufacturing operations where the production floor team may span multiple nationalities.
Yes. While BatchM has been designed with the rigour and traceability requirements of pharmaceutical manufacturing in mind, its core capabilities — dependency management, sequential process control, real-time multi-stream monitoring, operator assignment, automated reminders, and dynamic reporting — are directly applicable to any regulated, repetitive, or structured batch production environment. In food manufacturing, BatchM can manage the sequencing of multi-stage production runs — mixing, cooking, filling, labelling, and quality checks — with dependencies and deadlines defined across each step to ensure consistent, compliant production. In cosmetics manufacturing, BatchM supports the sequencing and monitoring of formulation, blending, and packaging operations across multiple product lines simultaneously, with full operator visibility and real-time supervisory oversight. In chemical processing, contract manufacturing, and nutraceutical production, BatchM’s structured process initiation, live monitoring, and completion reporting capabilities provide the operational discipline that regulated and quality-focused production environments require. For organisations managing batch operations across multiple product types or business units, BatchM’s multi-programme architecture allows all of these to be managed from a single platform — with dedicated batch registers for each product line or business unit and consolidated programme-level oversight from a single dashboard.
BatchM is a core module within the goPLIMS Integrated Project Execution Platform and is designed to connect with the broader suite to provide complete traceability from production initiation to close-out and beyond. Deviations or non-conformances identified during a batch — a process parameter outside specification, a raw material discrepancy, or an equipment performance issue — can be formally raised and managed in DiscrepM, with the deviation record directly linked to the batch in which it occurred. Changes to batch procedures, materials, or equipment arising from investigation outcomes can be formally managed in ChangeM, ensuring that every modification to the batch process is controlled, approved, and documented. Controlled documents referenced in batch execution — approved procedures, specifications, and method documents — are managed and version-controlled in EDocM, ensuring that operators always execute against the current approved documentation. Actions arising from batch completion reviews or deviation investigations can be tracked to closure in ActionR. For pharmaceutical organisations building a comprehensive digital production and quality management environment, BatchM is the operational execution layer that connects every batch event — every activity, every deviation, every change — to a governed, traceable, and inspection-ready record within the goPLIMS platform.
Yes, BatchM supports the digitalisation of deviation and exception management by enabling structured tracking and control of batch-related issues within a compliant, traceable system.
In pharmaceutical manufacturing and project environments, deviations and exceptions frequently occur during batch execution, documentation, and review processes. BatchM allows teams to capture these events in real time, link them directly to specific batches, and manage them through structured workflows.
Each batch-related deviation can be:
- Logged with full context, including batch reference and supporting data
- Assigned to responsible teams for investigation and resolution
- Tracked through its lifecycle with time-stamped updates
- Linked to actions, RFIs, or change controls where required
With role-based access control, only authorised personnel can access or modify batch-related information, ensuring data integrity and compliance with regulatory standards.
By digitising batch deviations and exception handling, BatchM eliminates manual tracking, reduces errors, and ensures that all issues are documented, resolved, and audit-ready—supporting inspection readiness and operational excellence.
Yes, BatchM supports knowledge management by capturing batch execution data, deviations, and resolutions in a structured and centralised system.
In pharma operations, each batch generates valuable knowledge—from process performance and deviations to corrective actions and outcomes. BatchM ensures this information is retained and organised, rather than being lost in paper records or disconnected systems.
This enables teams to:
- Analyse historical batch data and identify recurring issues
- Improve process consistency and reduce deviations over time
- Support continuous improvement and operational excellence
- Accelerate investigations using past batch insights
With searchable records, structured data capture, and role-based dashboards, BatchM makes it easy for teams to access relevant information while maintaining strict control over sensitive data.
By embedding knowledge capture into batch execution workflows, BatchM helps organisations move from reactive issue management to data-driven decision-making and continuous process improvement—a critical capability in regulated pharmaceutical environments.