Managing Pre-Manufacturing Phases with goPLIMS

Confidence, Control & Clarity
Product Development, Technology Transfer, Clinical Trials and Project Commissioning are the most complex, knowledge-intensive and externally dependent phases in the pharma lifecycle. goPLIMS connects all four in a single integrated platform — giving every team real-time visibility, structured execution and full traceability.
Pre-manufacturing phases are the hardest to manage — and the highest risk
External consultants, CROs, CMOs, multi-site teams and intense regulatory scrutiny make these phases uniquely complex. Most organisations manage them with tools built for a simpler world.
Fragmented Tools & Disconnected Teams
Engineering, CQV, quality, operations, R&D and regulatory teams each work in separate systems — emails, spreadsheets, SharePoint folders and vendor portals — with no single source of truth. Critical information is duplicated, conflicts are invisible and no one has a complete picture.
External Contributors Carry Critical Knowledge
Consultants, CROs, CMOs, equipment vendors and contractors hold irreplaceable tacit knowledge — process know-why, formulation rationale, site-specific experience. When their phase engagement ends, they leave. Without a governed platform, their knowledge leaves too.
Phase Transitions Treated as Milestones
The moment a phase completes — product development ends, tech transfer hands over, clinical close-out — is also the moment of maximum information loss. Without formal handover protocols & a shared platform, each transition is a knowledge cliff edge rather than a managed transfer.
Compliance & Audit Readiness Under Constant Pressure
GMP, ICH Q10, 21 CFR Part 11 — the regulatory requirements across pre-manufacturing phases are extensive and non-negotiable. Manual tracking of approvals, e-signatures and audit trails across disconnected systems creates compliance exposure at every step and inspection.
The majority of pharma projects are still run on spreadsheets, disconnected tools and emails. Teams spend their most productive hours in meetings, follow-ups and firefighting — not delivering. >30% of project team time estimated lost to avoidable coordination in manual environments, endless meetings and searching for information.
70%
$2.6B
80%
3×

Product Development
The foundation of every pharmaceutical product.
This phase generates the formulation knowledge, analytical methods, CMC data and process parameters that underpin everything that follows — and involves heavily external CRO and lab teams whose insights must be formally captured before phase completion.
- Key activities managed in goPLIMS integrated platform
-
Formulation decisions and process design rationale captured in DecisionM
-
CMC deviations & analytical method changes tracked in DiscrepM & ChangeM
-
Stability study actions and timelines managed in ActionR
-
CRO deliverables and milestone tracking via TrackR
-
Version-controlled protocols & reports stored in EDocM with full e-signature
-
Structured lessons learned captured at phase gate before CRO team transitions
and much more functions with other modules!
Technology Transfer
Technology transfer is where product knowledge must cross organisational and often geographical boundaries. The risk of losing the process understanding gained in development is highest here — where external CMOs, MS&T teams and receiving site QA must work together seamlessly across sites and time zones.
- Key activities managed in goPLIMS
-
Scale-up parameters and process equivalence decisions captured in DecisionM
-
Site-specific adaptations and batch deviations managed in DiscrepM
-
Transfer protocol changes governed with full approval routing in ChangeM
-
Cross-site team collaboration and action tracking in ActionR
-
CMO access to goPLIMS governed by role — their contributions captured before they exit
-
Transfer documentation controlled in EDocM with compliant e-signatures
and much more functions with other modules!


Clinical Trials
Clinical programmes involve the most complex stakeholder landscape in the entire pre-manufacturing lifecycle — CROs, clinical investigators, biostatisticians, regulatory writers and ethics bodies, often across multiple countries. Coordinating actions, amendments, safety signals and regulatory submissions demands a platform built for structured, governed execution.
- Key activities managed in goPLIMS
-
Protocol amendments and design decisions captured in DecisionM and ChangeM
-
Site deviations, safety signals and adverse events tracked in DiscrepM
-
Investigator actions and CRO deliverables managed in ActionR
-
Regulatory submission documents version-controlled in EDocM
-
Clinical close-out actions and CRO knowledge handover captured in goPLIMS before team exits
-
Real-time trial progress and milestone dashboards via TrackR
and much more functions with other modules!
Project Management & Commissioning & Qualification (C&Q)
Facility projects and C&Q programmes are among the most complex multi-stakeholder environments in pharma — engineering firms, equipment vendors, contractors, CQV consultants and internal quality teams all contributing across extended timelines. goPLIMS was built specifically for this environment by a founder with 30 years of pharma C&Q experience.
- Key activities managed in goPLIMS
-
Engineering design decisions and design intent captured in DecisionM
-
Construction NCRs and C&Q non-conformances managed in DiscrepM
-
Technical queries and vendor clarifications formalised in RFI-M
-
Commissioning defects tracked and closed in real time in PunchM
-
Change requests with full impact analysis and approval routing in ChangeM
-
IQ, OQ, PQ documentation and e-signatures managed in EDocM + ESignM
and much more functions with other modules!

What goPLIMS delivers across all four phases
One integrated platform replacing the patchwork of tools, emails and spreadsheets that currently manage your most critical work.
Real-Time Portfolio Visibility
Live dashboards across all 4 phases: phase progress, milestone status, action backlogs & deviation trends — without manual compilation or meetings. Leadership sees the full picture at any moment.
Structured Execution
Configurable workflows enforce the right process for every activity — change requests, deviation investigations, protocol amendments & vendor approvals all follow defined routes with no manual chasing.
Compliance Built In
21 CFR Part 11 compliant e-signatures, immutable audit trails % GxP-aligned workflows are core to goPLIMS architecture. Every record is inspection-ready from the moment it is created.
Governed Access
CROs, CMOs, contractors and consultants are given role-based access to goPLIMS — their deliverables captured, their actions tracked and their knowledge retained in the platform before they exit the engagement.
Structured Handovers
Formal knowledge & documentation handover is a built-in deliverable at every phase transition. Each phase gate closes fully complete, properly structured and record ready for the next team.
Knowledge Preserved
Decisions, deviations, changes & lessons learned from every phase are captured in a structured base — so teams can access pre-manufacturing history when investigations & CAPAs demand it.
Every module purpose-built for pre-manufacturing phase management
Each module works independently or as part of the fully integrated goPLIMS platform — configured to your phase workflows and scaled to your team size.
ActionR
Action Register Management
Centralise all team actions, tasks and meeting minutes. Kanban views, automated reminders and real-time closure tracking across all phases and teams.
DiscrepM
Discrepancy Management
Manage deviations, non-conformances and out-of-specification results with linked investigations, and compliant e-signature approval workflows.
ESignM
Electronic Signature Management
21 CFR Part 11 compliant e-signatures with unique control copies and full traceability — replacing paper-based approval processes across all pre-manufacturing phases.
ChangeM
Change Management
Govern all change requests with impact assessment, multi-level approval routing and full traceability — from protocol amendments to design modifications.
RFI-M
RFI Management
Centralised RFI log with full stakeholder accountability — formalise, track and close all technical queries from engineering firms, contractors and equipment vendors.
TrackR
Central Online Tracker
Real-time milestone and status dashboards — portfolio RAG status, phase progress and cross-team visibility for leadership and project teams without manual reporting.
DecisionM
Decision Management
Capture every critical decision with rationale, approver and context — preserving the institutional knowledge that underpins future investigations, and regulatory submissions.
PunchM
Punchlist & Defect Management
Track and close commissioning and construction defects in real time — on-site or remotely — with photographic proof, approval routing and full closure traceability.
EDocM
Electronic Document Management
Version-controlled, compliant document hub — protocols, reports, specifications and regulatory documents linked directly to the projects and deviations that generated them.
Knowledge Management
Every goPLIMS record is simultaneously a structured knowledge asset — decisions, deviations and changes captured during pre-manufacturing phases form a searchable knowledge base for manufacturing operations. For a dedicated overview of goPLIMS Knowledge Management capabilities visit our KM page

5-tier structure
Every record contextualised & governed
The fundamental failure of pre-manufacturing phase management is not a lack of information — it is a lack of structure. Emails, SharePoint folders and vendor portals contain enormous volumes of data. The problem is that none of it is connected, classified or searchable across phase boundaries.
goPLIMS solves this by applying a consistent five-tier taxonomy to every record — regardless of which module created it, which team owns it or which phase it belongs to.
When a manufacturing team investigates a deviation, they can search across all pre-manufacturing phases — finding the relevant product development decision, technology transfer deviation or commissioning non-conformance — in seconds, not days.
That is the operational difference between a document system and an integrated execution platform.
goPLIMS provides a structured digital framework that connects modules, programs, workstreams and governance layers into a single controlled platform. From action registers and RFIs through to departmental ownership, access control and taxonomy management, every record is linked, traceable and searchable across the entire project lifecycle. This unified structure ensures teams, contractors and stakeholders operate within a governed environment — improving collaboration, maintaining compliance and delivering complete operational visibility from design through commissioning.
Built for regulated pre-manufacturing environments
goPLIMS is designed from the ground up for regulated pharma environments — with compliance woven into every workflow, not added as an afterthought.
What inspection-readiness looks like with goPLIMS
Every decision logged — with rationale, approver identity and timestamp, retrievable in seconds across any phase
No paper trail gaps — all approvals, changes and deviations captured in a single digital record with no reliance on emails
GxP workflows enforced — no one can complete an activity without the correct approval sequence in place
External access governed — CRO, CMO and contractor contributions tracked and traceable even after their engagement ends
Phase gate records complete — every handover includes a structured, auditable record of what was done, decided and transferred
Inspection-ready from day one — evidence is structured, searchable and exportable at any point during execution, not only at audit time

Measurable outcomes from your first phase
40%
Reduction in phase reporting time — live dashboards replace manual status compilation and update meetings
25%
Improvement in on-time phase delivery through structured action management and real-time escalation
60%
Faster deviation resolution — prior phase knowledge accessible at point of investigation, not buried in email
35%
Reduction in external contributor management overhead through governed platform access and automated tracking
50%
Less time in status meetings — teams stay aligned through live dashboards, not weekly update cycles
100%
Audit trail coverage — every action, decision, change and approval logged automatically from phase start
The cost of poor pre-manufacturing phase management
- Phase overruns and scope creep → delayed product launches → revenue and competitive loss
- Poor tech transfer documentation → scale-up failures → batch losses and repeat investigations
- External contributor knowledge lost at engagement close → extended onboarding for next team
- Manual compliance tracking → audit trail gaps → regulatory findings and remediation cost
- Disconnected change control → as-built deviations from approved design → requalification overhead
- No phase gate handover → manufacturing teams inherit assets without context → operational risk
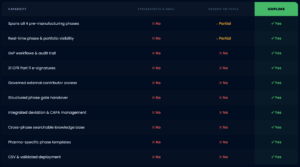
Why pharma teams choose goPLIMS for pre-manufacturing phases
See how goPLIMS compares to the tools most teams currently use to manage their most complex phases.
One platform for all your pre-manufacturing phases.
Connect your Product Development, Technology Transfer, Clinical Trials and C&Q programmes in goPLIMS — and give every team the visibility, structure and compliance they need to deliver.