One Platform. Every Workflow. Clarity. Confidence. Control.
Discover how goPLIMS — the Integrated Project Execution & Management Platform — unifies every workflow, programme, project, and workstream in a single execution layer, giving pharma and regulated industry teams proactive control from design and construction through to manufacturing readiness.
The Challenge of Complex Project Execution in Pharma
Regulated industry projects — whether a new manufacturing facility, a process equipment upgrade, or a clinical trial programme — are among the most complex programmes any organisation undertakes. They span years, involve dozens of stakeholders, require meticulous documentation, and operate under strict regulatory scrutiny where missed actions, undocumented decisions, and uncontrolled changes carry real compliance risk.
The traditional response to this complexity has been to layer on more tools — a separate system for documents, another for actions, another for changes, another for meeting minutes, another for punchlists. The result is not simplicity. It is fragmentation.
The Hidden Cost of Fragmentation
Disconnected Communication
Actions agreed in meetings never make it into the tracker. Decisions get buried in email chains. Teams operate on different versions of the truth.
Compliance Risk
Undocumented deviations, informal change approvals, and missing audit trails create regulatory exposure that is expensive to remediate after the fact.
Reactive Rather Than Proactive
Without a unified real-time view, managers find out about problems after they have escalated — when intervention is costly rather than routine.
Delays and Cost Overruns
Coordination overhead, duplicated effort, missed handovers, and bottlenecks that go undetected until they cascade — all compounding into schedule and budget impact.
What is goPLIMS?
goPLIMS is an Integrated Project Execution & Management Platform — purpose-built for pharma and regulated industries. It provides a single execution layer that integrates multiple workflows, programmes, projects, and workstreams, connecting documents, actions, punchlists, deviations, changes, decisions, RFIs, and more in one unified platform.
Rather than replacing existing business processes, goPLIMS digitises and connects them — giving every team member, from field executor to project director, exactly the tools, visibility, and governance they need to perform their role with confidence. The platform delivers real-time visibility, workflow governance, and compliance-ready execution across the entire project lifecycle, helping organisations reduce coordination time by up to 50% and accelerate speed to market.
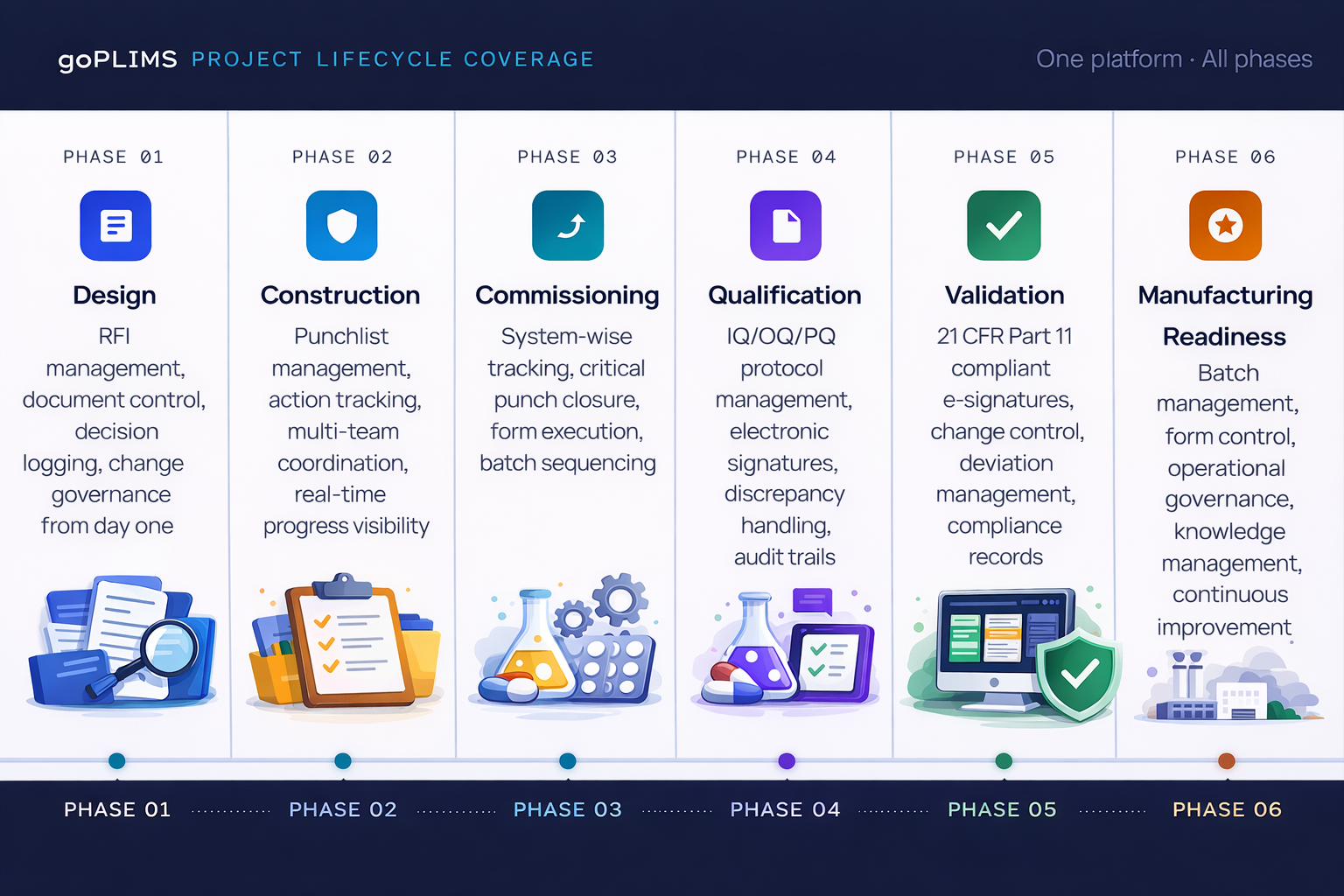
Full Lifecycle Coverage — Design to Manufacturing Readiness
Unlike point solutions that address a single phase or function, goPLIMS is designed to accompany teams across the entire project lifecycle — from the earliest design decisions through to validated, manufacturing-ready operations:
The Single Execution Layer — How goPLIMS Connects Everything
The defining characteristic of goPLIMS is not the breadth of its individual modules — it is the way they work together as a single, integrated execution layer. Every workflow connects to every other workflow, creating a coherent, searchable, and auditable record of the entire project:
Actions Connect to Every Workflow
Every RFI, punchlist item, discrepancy, change request, and decision can generate linked actions in ActionR — creating an unbroken thread of accountability from first identification through to closure, regardless of which module originated the task.
Documents Integrate Across All Modules
EDocM provides the central, access-controlled document repository for the entire platform — meaning that forms completed in FormM, signed records from ESignM, and attachments from ChangeM or DiscrepM all land in one governed, searchable location.
Electronic Signatures Apply Platform-Wide
ESignM’s 21 CFR Part 11 compliant e-signature framework is available across ChangeM, DiscrepM, FormM, and beyond — so any workflow requiring formal approval benefits from the same authenticated, auditable signature system.
TrackR Provides the Real-Time Oversight Layer
While individual modules manage their own workflows, TrackR surfaces a unified, traffic-light-coded view of overall project health — giving project managers and senior leadership a single dashboard that spans every active workstream simultaneously.
Decisions Contextualise Every Outcome
DecisionM links every key project decision to the projects, risks, and workstreams it affects — so that when a change is made, a deviation resolved, or an RFI answered, the underlying rationale and accountable owner are permanently traceable.
Meeting Management Closes the Loop
ActionR’s built-in Meeting Management module ensures that every decision, action, and agreement made in a project meeting is immediately live in the platform — with smart, role-based minutes automatically distributed to every team member, keeping the platform and the meeting room perfectly synchronised.
The goPLIMS Module Ecosystem
goPLIMS delivers its integrated execution layer through a comprehensive suite of purpose-built modules — each addressing a specific workflow, each natively connected to the others:
RFI-M
RFI Management
PunchM
Punchlist Management
ChangeM
Change Management
DiscrepM
Discrepancy Management
EDocM
Document Management
ESignM
E-Signature Management
FormM
Form Management
BatchM
Batch Management
DecisionM
Decision Management
TrackR
Central Tracker
ActionR
Action Register
Everything goPLIMS delivers can be distilled into three outcomes that matter most to project managers, programme directors, and regulated industry leadership:
CLARITY
When every workflow lives in one platform, there is no ambiguity about what is happening, who owns it, or where it stands.
- Real-time status of every action, document, and workstream
- Traffic light dashboards giving leadership the big picture instantly
- Role-based views ensuring every team member sees exactly what they need
- A single source of truth — no conflicting versions, no stale reports
CONFIDENCE
Full compliance, traceability, and audit-readiness — always. Not just at inspection time, but every single day of the project.
- 21 CFR Part 11 compliant digital signatures across all approvals
- Complete, tamper-evident audit trails for every action and decision
- Formal governance workflows that eliminate uncontrolled change
- Documentation integrity from initiation through to final closure
CONTROL
Proactive management of complex programmes — identifying issues before they escalate and keeping every team focused on delivering outcomes.
- Bottleneck identification and resolution in real time
- Automated notifications ensuring nothing is missed or delayed
- Named accountability on every action, change, and decision
- Continuous improvement driven by platform-wide historical data
goPLIMS reduces coordination time by up to 50%
not by removing necessary governance, but by making governance effortless. When every workflow lives in one integrated platform, the overhead of managing complexity disappears. What remains is pure execution.
Measurable Outcomes Across the Programme
Teams that deploy goPLIMS consistently report measurable improvements across every dimension of project performance:
Reduction in Coordination Time
Automated workflows, integrated modules, and real-time visibility eliminate the overhead of chasing status, distributing minutes, and reconciling disconnected systems.
Single Source of Truth
One platform replaces dozens of disconnected tools — eliminating version conflicts, missed updates, and the compliance risk of undocumented decisions and uncontrolled changes.
Compliance Gaps at Inspection
Complete, tamper-evident audit trails, formal digital sign-off at every stage, and integrated documentation mean teams are inspection-ready at every moment — not just at audit time.
Global Access & Visibility
Remote and globally distributed teams access the same live platform from any device, any time zone — keeping global programmes coordinated without time-zone-driven delays.
Why Teams Choose goPLIMS
Replace multiple disconnected tools with one integrated execution platform that covers every project workflow
Manage complex, multi-phase programs proactively — identifying & resolving issues before they become schedule or compliance events
Deliver end-to-end compliance and audit-readiness — from design through to manufacturing readiness — without additional overhead
Give every stakeholder — from field executor to project director — the right information, at the right level of detail, at the right time
Purpose-built for pharma and regulated industries — not a generic project tool adapted for compliance, but a platform designed for it from the ground up
Accelerate speed to market by reducing coordination time by up to 50% — converting governance overhead into competitive advantage
About goPLIMS
goPLIMS is an Integrated Project Execution & Management Platform — delivering real-time visibility, workflow governance, and compliance-ready execution across the entire project lifecycle. Purpose-built for pharma and regulated industries, goPLIMS reduces coordination time by up to 50%, helping teams accelerate speed to market.
One Platform. Every Workflow. Total Control.
Discover how goPLIMS can transform your project execution — from design through to manufacturing readiness — with clarity, confidence, and control.